Proton
A proton is part of an atom.[1] They are found in the nucleus of an atom along with neutrons.[1] The periodic table groups atoms according to how many protons they have. A single atom of hydrogen (the lightest kind of atom) is made up of an electron moving around a proton. Most of the mass of this atom is in the proton, which is almost 2000 times heavier than the electron. Protons and neutrons make up the nucleus of every other kind of atom. In any one element, the number of protons is always the same. Scientists discovered in the early 20th century that an atom's atomic number is equal to the number of protons in that atom.
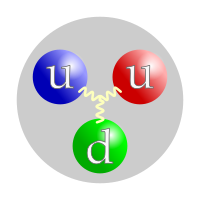
A proton is made up of 3 quarks,[1] two up quarks and one down quark.[1] One down quark has a charge of -1/3, and two up quarks have a charge of +2/3 each. This adds to a charge of +1. A proton has a very small mass. The mass of the proton is about one atomic mass unit. The mass of the neutron is also about one atomic mass unit. The size of a proton is determined by the vibration of the quarks that are in it, and these quarks effectively form a cloud. This means that a proton is not so much a hard ball as an area that contains quarks.
Related pages
changeReferences
change- ↑ 1.0 1.1 1.2 1.3 Cox, Brian; Cohen, Andrew (2011). Wonders of the Universe. HarperCollins. p. 108-109. ISBN 9780007395828.