Cytochrome
Cytochromes are haemoproteins (or proteins containing haem groups). They make ATP by electron transport.
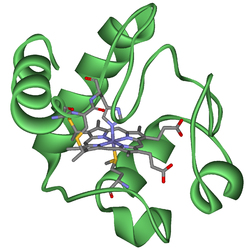
They are found either as single (monomeric) proteins (e.g., cytochrome c) or as part of bigger enzyme complexes which catalyze redox reactions.
The haem group does the redox reactions. These include all chemical reactions in which atoms have their oxidation state changed. In general, redox reactions involve the transfer of electrons.
History
changeCytochromes were described in 1884 as respiratory pigments. They include (haemoglobin, myoglobin, haemocyanin, haemoerythrin and chlorocruorin.[1][2] In the 1920s, Keilin rediscovered these respiratory pigments. He called them the cytochromes, or “cellular pigments”, and classified them as haem proteins.
Types
changeSeveral kinds of cytochrome exist and can be distinguished by spectroscopy. Three types of cytochrome are distinguished by their prosthetic groups.[3] In mitochondria and chloroplasts, these cytochromes are often combined in electron transport and related metabolic pathways.
References
change- ↑ A respiratory pigment increases the oxygen-carrying ability of the blood.
- ↑ MacMunn C.A. (1886). "Researches on myohaematin and the histohaematins". Philosophical Transactions of the Royal Society of London. 177. The Royal Society: 267–298. doi:10.1098/rstl.1886.0007. JSTOR 109482. S2CID 110335335.
- ↑ Prosthetic group = add-on bit which makes it work